Oxidation And Reduction :
Definitions :
i) Oxidation = process which involve gaining of oxygen (formation of an oxide)
ii) Reduction = process which involve losing of oxygen (decomposition of oxide)
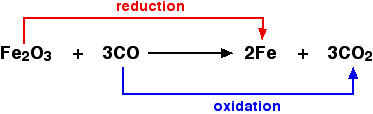
The equation below shows the example between oxidation and reduction process :
i) Oxidation = process which involve gaining of oxygen (formation of an oxide)
ii) Reduction = process which involve losing of oxygen (decomposition of oxide)
The equation below shows the example between oxidation and reduction process :
The reaction is both involving reduction and oxidation processes. If both process are going side-by-side, this process is called redox reaction.
Redox reaction involed :
i) Reducing Agent : Remove oxygen from another substance
ii) Oxidation Agent : Adding/ giving oxygen to another substance
In the reaction, the iron (III) oxide is the reducing agent because it loss of oxygen and the carbon dioxide is the oxidation agent as it giving an oxygen.
Oxidation & Reduction in Hydrogen Transfer :
Oxidation = loss of hydrogen
Reduction = gain of hydrogen
Oxidation & Reduction in Electron Transfer :
Oxidation = loss of electrons
Reduction = gain of electrons
OIL RIG
Oxidation is loss Reduction is gain
SUMMARY :
* Oxidation : gain of OXYGEN, loss of HYDROGEN, loss of ELECTRONS
* Reduction : loss of OXYGEN, gain of HYDROGEN, gain of ELECTRONS
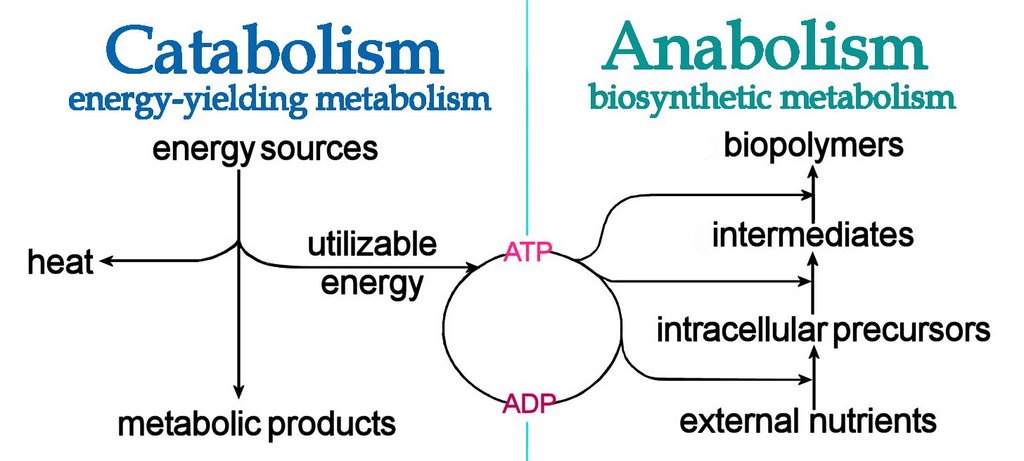
The Importance of redox reactions in metabolism :
Redox reaction involed :
i) Reducing Agent : Remove oxygen from another substance
ii) Oxidation Agent : Adding/ giving oxygen to another substance
In the reaction, the iron (III) oxide is the reducing agent because it loss of oxygen and the carbon dioxide is the oxidation agent as it giving an oxygen.
Oxidation & Reduction in Hydrogen Transfer :
Oxidation = loss of hydrogen
Reduction = gain of hydrogen
Oxidation & Reduction in Electron Transfer :
Oxidation = loss of electrons
Reduction = gain of electrons
OIL RIG
Oxidation is loss Reduction is gain
SUMMARY :
* Oxidation : gain of OXYGEN, loss of HYDROGEN, loss of ELECTRONS
* Reduction : loss of OXYGEN, gain of HYDROGEN, gain of ELECTRONS
The Importance of redox reactions in metabolism :
Metabolism can further broken down into two subgroups which are anabolism and catabolism:
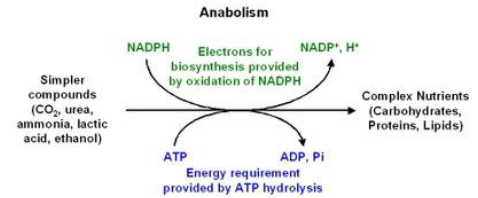
ANABOLISM :
- refers to the chemical reactions where the simpler substances are combined to form more complex molecules.
- usually require energy to build new molecules and/or store energy.
Human can synthesize amino acid by anabolism from the available metabolites by glycolysis, TCA cycle and pentose phosphate pathway by the nitrogen-carrier molecules :
GLUTAMATE or GLUTAMINE
HOW ???
i) Direct incorporation of ammonium ion into organic molecules
* Amino acids are synthesized by incorporating free ammonium ion into organic molecules.
- Amination (product = Glutamate)
- Amidation (product = Glutamine)
ii) Transamination reaction
* Amino groups are transferred from molecules to molecules :
- Glutamate = an amino groups donor
- Glutamine = an amino groups acceptor
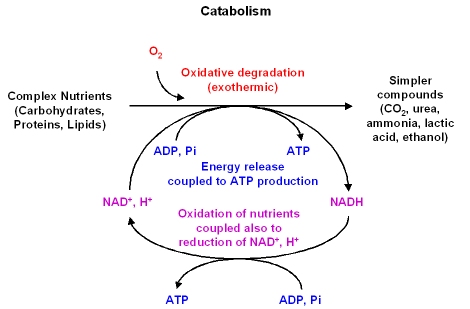
CATABOLISM :
Important :
- As metabolic fuels
- Removal of nitrogen
- Eliminate the excess nitrogen
This chemical reaction remove of the nitrogen in the amino acids that involve by 2 stages:
i) Deamidation = Remove one nitrogen from the R-group of an amino acid
ii) Transamination = Remove the amino group in an amino acid
The implication of this reaction are: amino group (from original amino acids) is transferred to α-ketoglutarate and glutamate as the product.
ANABOLISM :
- refers to the chemical reactions where the simpler substances are combined to form more complex molecules.
- usually require energy to build new molecules and/or store energy.
Human can synthesize amino acid by anabolism from the available metabolites by glycolysis, TCA cycle and pentose phosphate pathway by the nitrogen-carrier molecules :
GLUTAMATE or GLUTAMINE
HOW ???
i) Direct incorporation of ammonium ion into organic molecules
* Amino acids are synthesized by incorporating free ammonium ion into organic molecules.
- Amination (product = Glutamate)
- Amidation (product = Glutamine)
ii) Transamination reaction
* Amino groups are transferred from molecules to molecules :
- Glutamate = an amino groups donor
- Glutamine = an amino groups acceptor
CATABOLISM :
Important :
- As metabolic fuels
- Removal of nitrogen
- Eliminate the excess nitrogen
This chemical reaction remove of the nitrogen in the amino acids that involve by 2 stages:
i) Deamidation = Remove one nitrogen from the R-group of an amino acid
ii) Transamination = Remove the amino group in an amino acid
The implication of this reaction are: amino group (from original amino acids) is transferred to α-ketoglutarate and glutamate as the product.