PROTEINS :
Protein is the macromolecule and it is an organic compound that composed of one or more long chains of amino acids which bonded together.
The example of protein that we can get from our diet such as fish, meat and peanut.
There are different classes of amino acids:
a) Non-polar amino acids (Alkyl and Aromatic groups)
- Glycerine, Valine, Leucine, Methionine, Alanine,Isoleucine, Proline, Tryptophan, Phenylalanine
b) Polar amino acids (Neutral, Basic and Acidic)
-Serine, Threonine, Tyrosine, Cysteine, Asparagine, Glutamine
c) Acidic amino acids
-Aspartic, Glutamic
d) Base amino acids
-Histidine, Lysine, Arginine
* Levels of Protein Organization:
The protein levels can divided into 4 structure:
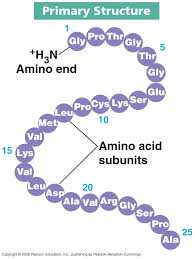
a) Primary structure:
-Only the sequence of amino acids in the protein.
a) Primary structure:
-Only the sequence of amino acids in the protein.
b) Secondary structure:
- α helices and β pleated sheets is formed together by the hydrogen bond between the peptide bond.
- α helices and β pleated sheets is formed together by the hydrogen bond between the peptide bond.
c) Tertiary structure:
- The alpha helices and beta pleated sheets are folding together by the R-groups.
- The alpha helices and beta pleated sheets are folding together by the R-groups.
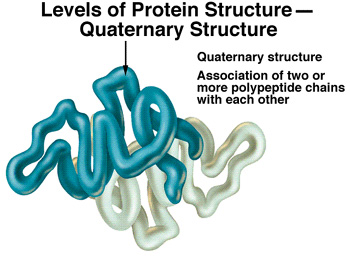
d) Quaternary structure:
- More than one polypeptide are associate together to form protein complex which influenced by the R-groups.
- More than one polypeptide are associate together to form protein complex which influenced by the R-groups.
REMINDER !!!
* AMINO ACID = basic units of protein.
* PEPTIDE = composed of 2 or more amino acids.
* POLYPEPTIDE = 10 or more amino acids.
These amino acids will form a protein when they are bonded together.
* AMINO ACID = basic units of protein.
* PEPTIDE = composed of 2 or more amino acids.
* POLYPEPTIDE = 10 or more amino acids.
These amino acids will form a protein when they are bonded together.
|
In tertiary structure, there are several factor that influenced:
i) Hydrophobic/Hydrophilic interactions ii) Hydrogen bonding iii) Disulfide linkages iv) Chaperone proteins folded In the quaternary structure, the factors that influenced the structures i) Hydrophobic/Hydrophilic interactions ii) Hydrogen bonding ii) The shape and charge distribution on associating polypeptides. *Classification by Protein Structure:1. Conjugated proteins contain non-amino acids groups.
2. Amino acid portion called apoprotein and non-amino acid portion is called prosthetic group. |
*Type of Protein and Function:
1. Structural protein
For the structure support
2. Enzymatic protein
Selective acceleration of chemical reactions
3. Hormonal protein
Coordination of an organism activities
4. Receptor protein
The response of the cell towards the chemical stimuli
5. Transport protein
Transport the other substances
6. Contractile and motor proteins
Help in the movement of protein
7. Defensive protein
For the protection against the diseases
For the structure support
2. Enzymatic protein
Selective acceleration of chemical reactions
3. Hormonal protein
Coordination of an organism activities
4. Receptor protein
The response of the cell towards the chemical stimuli
5. Transport protein
Transport the other substances
6. Contractile and motor proteins
Help in the movement of protein
7. Defensive protein
For the protection against the diseases
*Classification of Amino Acids:
*Function of Amino Acids:
a) Acts as a synthesize neurotransmitter
Example: Tryptophan and Tyrosine
- Tryptophan : produce serotonin, which regulates our body and makes the hormone melatonin.
- Tyrosine : Used to synthesis norepinephrine and adrenaline
Both of this hormone compete with each other for access to our brain.
b) Metabolized for energy in body
Involved : glutamic acid, cysteine and glycine that combine to form glutathione (antioxidant)
c) Protect cardiovascular health
Our body uses arginine to make nitric oxide.
Nitric oxide: helps in lower the blood pressure by relaxing the muscle in blood vessels and produced in heart muscle, where it regulate muscles.
d) Build up protein
DNA define specific amino acids and they must connect to build the protein.
Example: Tryptophan and Tyrosine
- Tryptophan : produce serotonin, which regulates our body and makes the hormone melatonin.
- Tyrosine : Used to synthesis norepinephrine and adrenaline
Both of this hormone compete with each other for access to our brain.
b) Metabolized for energy in body
Involved : glutamic acid, cysteine and glycine that combine to form glutathione (antioxidant)
c) Protect cardiovascular health
Our body uses arginine to make nitric oxide.
Nitric oxide: helps in lower the blood pressure by relaxing the muscle in blood vessels and produced in heart muscle, where it regulate muscles.
d) Build up protein
DNA define specific amino acids and they must connect to build the protein.
*Anabolism vs. Catabolism Amino Acids:
(ANABOLISM AMINO ACIDS)
|
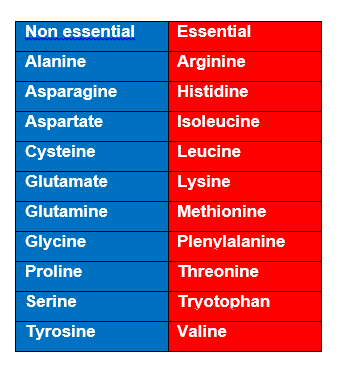
Amino acids are differentiate into two categories:
1. Non essential : Human can synthesize from the available metabolites. Can produce instantly. 2. Essential : Cannot be produced by human. Human must take from the food source. BUT, for Arginine, Methionine and Phenylalanine they are not directly related to lack of synthesis.
ARGININE : synthesized by mammalian cells but a rate that insufficient to meet the growth needs the body and the majority that is synthesized is cleaved to form urea. METHIONINE : Required in large amounts to produce cysteine if the latter amino acids is not adequately supplied in the diet. PHENYLALANINE : Needed in large amounts to form tyrosine if the latter is not adequately supplied in the diet. |
Human can synthesize amino acids from intermediates of :
1. Glycolysis
2. TCA cycle
3. Pentose phosphate pathway
Although amino acids must have amino group, it can be obtained from the nitrogen-carrier molecules which are the GLUTAMATE or GLUTAMINE.
*Glutamate is the neurotransmitter in brain.
*Glutamate without carboxylate group is GABA (major inhibitory neurotransmitter in brain).
1. Glycolysis
2. TCA cycle
3. Pentose phosphate pathway
Although amino acids must have amino group, it can be obtained from the nitrogen-carrier molecules which are the GLUTAMATE or GLUTAMINE.
*Glutamate is the neurotransmitter in brain.
*Glutamate without carboxylate group is GABA (major inhibitory neurotransmitter in brain).
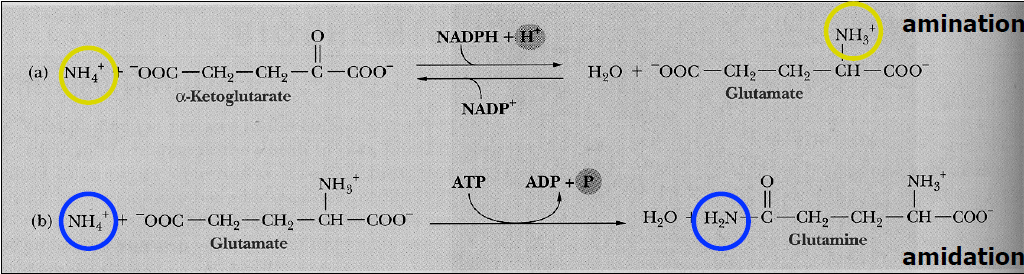
*Ways To Biosynthesized Amino Acids:
1. Direct incorporation of ammonium ion into the organic molecules:
a) Amination
- Creation of an amine, either by addition of an amino group to an organic compound or by reduction of nitro compound.
b) Amidation
- The reaction with or forming of an amide.
a) Amination
- Creation of an amine, either by addition of an amino group to an organic compound or by reduction of nitro compound.
b) Amidation
- The reaction with or forming of an amide.
(CATABOLISM AMINO ACIDS)
|
Catabolism of amino acids are important:
i) As metabolic fuels ii) Removal of nitrogen iii)Eliminate the excess nitrogen Amino acids can catabolize in the liver from: 1. Diet 2. Biosynthetic reactions 3. Normal turnover of intracellular proteins The organ that can undergo deamination :
1. LIVER - Synthesis plasma proteins - Involve gluconeogenesis, ketogenesis, uncatabolized branched chain of amino acids and synthesis of urea 2. KIDNEY - Function : To metabolized glutamine into α-ketoglutarate (for gluconeogenesis) + NH4 - NH4 that produced from the arginine synthesis is excreted for the urea cycle to preventing acidosis in buffer. 3. SKELETAL MUSCLE - Synthesis the muscle protein - Alanine is catabolized to the liver for gluconeogenesis, whereas the glutamine is metabolized to kidneys |
WAYS TO REMOVE THE AMINO ACIDS
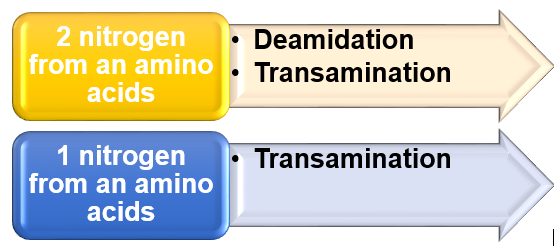
*Deamination is the process to remove 1 nitrogen from the R-group of an amino acid
-Example : In liver glutamate undergoes oxidative deamination by help of glutamate dehydrogenase. *Transamination is the process to remove the amino group in an amino acid. ^^^PRODUCT : Produce glutamate by transferring the amino group to α-ketoglutarate ^^^ |